Supercooling
|
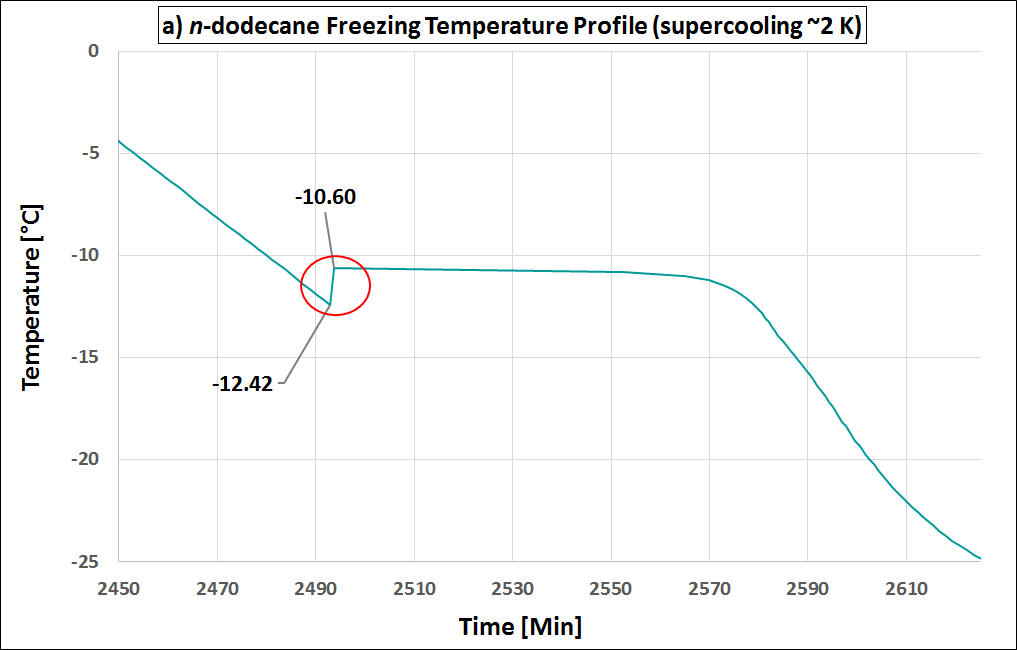
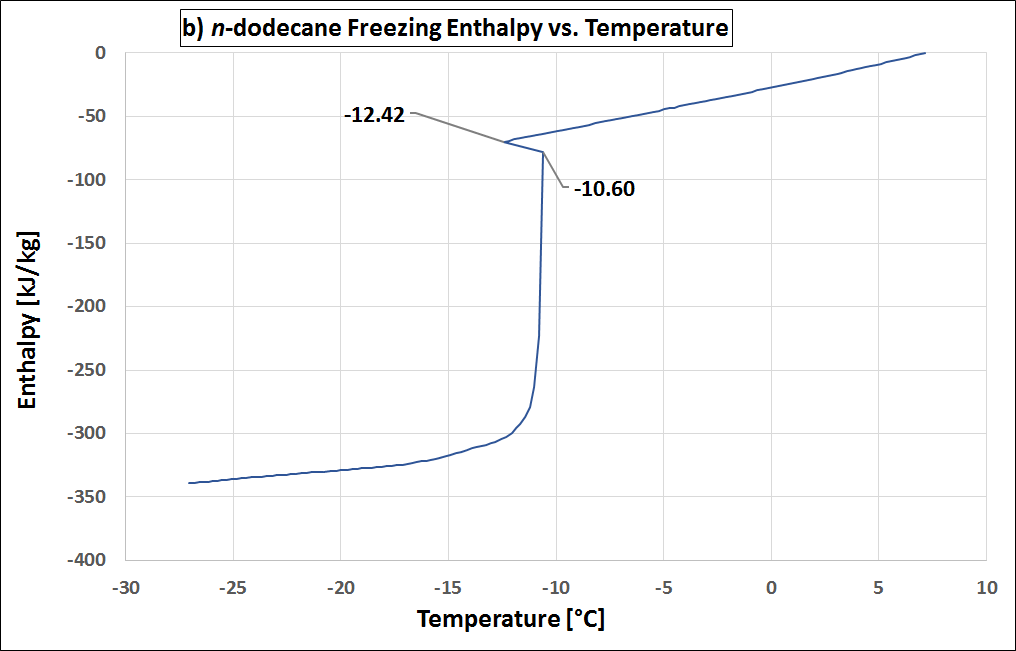
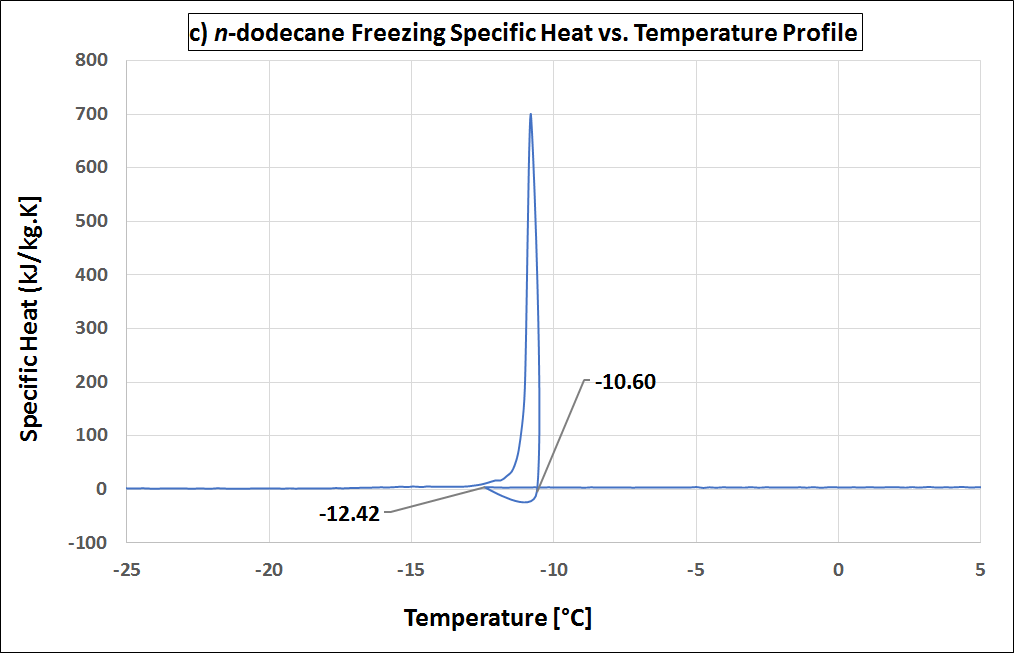
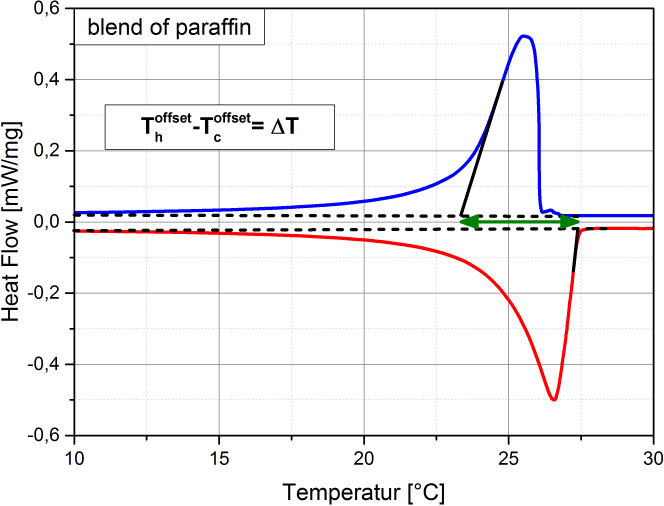
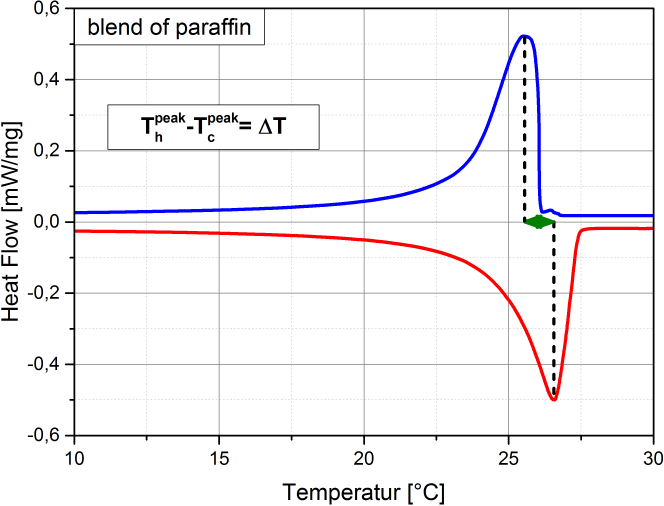
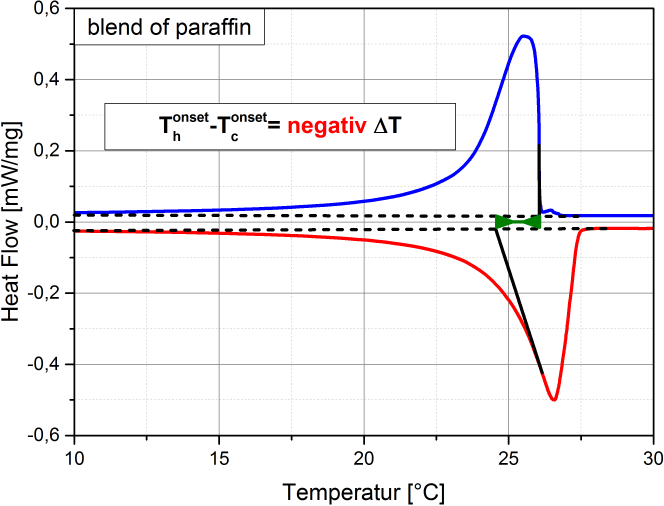
Supercooling is the process of cooling a liquid or a gas below its freezing point without it becoming a solid. (see also Wikipedia). Supercooling during the liquid-solid phase change is the phenomenon when a material’s crystallization initiation occurs at a temperature below its freezing temperature. That is, the nucleation starts at a temperature below the real freezing point of the material. Thus, a material that tends to supercool should be cooled considerably below its expected freezing point to initiate freezing. Once its nucleation initiates, the material temperature rises to its real freezing point, and then continues freezing at that temperature. If supercooling is considerable, it is disadvantageous in a PCM as it negatively affects the functionality of the TES system. That is because, then the freezing does not start at the expected freezing temperature, but only way below, with a practical requirement of a large operating temperature range. In addition, the higher the degree of supercooling, the less the energy released (as it is released as sensible heat, which is lesser than the expected latent heat release). Many materials undergo supercooling. Supercooling is related to the material’s nucleation and solidification mechanisms. The degree of its supercooling is influenced by the sample size, homogeneity, cooling rate and sample container surface morphology (influencing the availability of nucleation sites), among other factors. A pure (i.e., unary) material which undergoes supercooling, will rise-up to its real freezing temperature and then will freeze within a narrow temperature range (ideally: at constant temperature). Figure 1 is an example of a pure material: n-dodecane (purity: 99%), that supercools. In Figure 1 (a), the supercooling region is enclosed by a red circle. When the temperature is -12.42 °C the nucleation starts. Right after, the temperature rises back to its real freezing point around -10.6 °C and remains rather constant around this temperature till freezing completion. The supercooling is characterized also in the enthalpy and specific heat profiles of n-dodecane, as in Figure 1 (b) and (c) respectively, indicated with the crystallization initiation and the real freezing (initial) temperatures. Blends could also undergo supercooling. Supercooling in blends will cause phase separation, if these are incongruent melting compositions. Only congruent melting compositions will not phase separate even with supercooling. Peritectic compounds (they are a type of incongruent melting compositions) will inherently supercool, as their solidification occurs through coring. Coring is a process where each solidifying block forms a core with a higher melting point surrounded by outer layers of increasingly lower melting points. To initiate this core formation, the material should be cooled below its real freezing point. As peritectics will always supercool, they will always phase separate, and therefore are unsuitable as PCMs. If a eutectic supercools, it will also have phase separation. That is because a eutectic is not a congruent melting blend. Therefore eutectics are suitable as PCMs only in the absence of supercooling. That is because the eutectic solidification (of all the involved components) occurs simultaneously (if supercooling is absent), such that their total composition is the same as the composition of the liquid. Therefore, if there is a eutectic that does not undergo supercooling, it freezes rather similar to a congruent melting composition, and is thus suitable as a PCM. How is supercooling determined: For very pure and homogeneous substances, supercooling is determined by the difference between the onset melting temperature and the nucleation temperature. It should be noted here that the nucleation is a stochastic process which, in contrast to the melting point, depends on the measuring conditions (e. g. cooling rate, sample volume, etc.). Therefore the measured value referes to these settings. This definition is not applicable to PCMs that do not show a nucleation temperature such as blends of paraffins. These materials show not a clear nucleation temperature, the behaviour is more like a hysteresis. Figure 2 - figure 4 show different approaches to determine the value for supercooling of RT27. Reference points are: peak-peak, offset-offset and onset-onset temperature. Peak-peak (figure 3) and offset-offset (figure 4) are very much dependent on the heating rate, sample mass and the DSC device. Therefore, these differences are only comparable within the same DSC if the same heating rate and approximately the same thermal mass have been used. As the difference between onset temperature of the melting peak and crystallisation peak can be negative this is not an indicator for the degree of supercooling. The challenge with blends or other inhomogeneous materials is that they have a melting and crystallization. So no nucleation point is visible. |
Figure 1: Supercooling in pure materials: e.g. n-dodecane, as seen in its (a) temperature-profile, (b) enthalpy profile and (c) specific heat profile. Figure 2: Possibility offset-offset temperature Figure 3: Possibility peak-peak temperature Figure 4: Possibility onset-onset temperature |
|---|